Unit 7 – Electrochemistry (Short Questions)
Q.1 Define oxidation in terms of electrons. Give an example.
Q.2 Define reduction in terms of loss or gain of oxygen or hydrogen. Give an example.
Q.3 What is difference between valency and oxidation state?
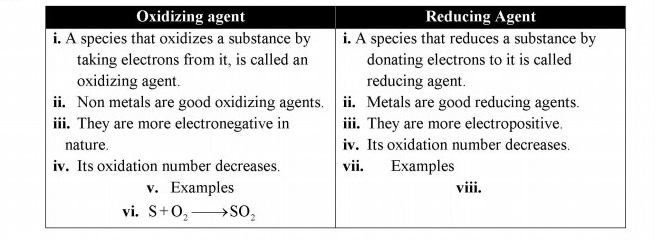
Q.4 Differentiate between oxidizing and reducing agents
Q.5 Differentiate between strong and weak electrolytes.
Q.6 How electroplating of tin on steel is carried out?
Q.7 Why steel is plated with nickel before the electroplating of chromium.
Q.8 How can you explain, that following reaction is oxidation in terms of increase of oxidation number Al” Al° — A1+3 + 3e–
Q.9 How can you prove so it is an oxidation reaction with an example that conversion of an ion to an atom is an oxidation process?
Q.10 Why does the anode carries negative charge in galvanic cell but positive charge in electrolytic cell? Justify with comments.
Q.11 Where do the electrons flow from Zn electrode in Daniel’s cell?
Q.12 Why do electrodes get their names ‘anodel and cathode in galvanic cell?
Q.13 What happens at the cathode in a galvanic cell?
Q.14 Which solution is used as an electrolyte in Nelson’s cell?
Q.15 Name the by-products produced in Nelson’s cell?
Q.16 Why galvanizing is done?
Q.17 Why an iron grill is painted frequently?
Q.18 Why O2 is necessary for rusting?
Q.19 In electroplating of chromium, which salt is used as an electrolyte?
Q.20 Write the redox reaction taking place during the electroplating of chromium?
Q.21 In electroplating of silver, from where Ag + come and where they deposit?
Q.22 What is the nature of electrode used in electroplating of chromium?
Q.1 Define oxidation in terms of electrons. Give an example.
Answer:
Oxidation is the loss of electron by an atom or an ion e.g,

Q.2 Define reduction in terms of loss or gain of oxygen or hydrogen. Give an example.
Answer:
Reduction:
“The addition of hydrogen or removal of oxygen during a chemical reaction.”
Examples:
 i. ZnO + C → 2Zn + CO2
i. ZnO + C → 2Zn + CO2
ii. H2 + Cl2 → 2HCl
Q.3 What is difference between valency and oxidation state?
Answer:
| Valency | Oxidation Number or state |
|
|
|
|
|
|
Q.4 Differentiate between oxidizing and reducing agents
Answer:
Q.5 Differentiate between strong and weak electrolytes.
Answer:
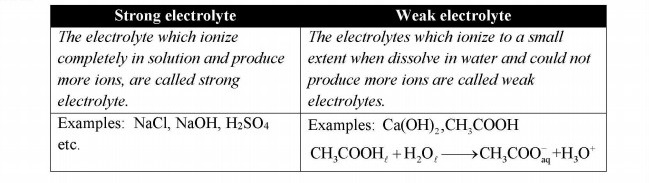
Q.6 How electroplating of tin on steel is carried out?
Answer:
In electroplating of silver, when current is passed through the cell. A.g. ions present in the electrolyte solution migrate towards the cathode and deposit after picking up electrons. The anode consists of silver bar or sheet. Which is oxidized to Ag ions which dissolve in solution and migrate towards the cathode where they are discharged and deposited on the object
At anode: Ag(8) → Ag+(aq) + e–
At cathode: Ag (aq) → Ag(8)
Q.7 Why steel is plated with nickel before the electroplating of chromium.
Answer:
The steel is usually plated first with nickel or copper then by chromium because it does not adhere well on the steel surface. Moreover, it allows moisture to pass through it and metal is stripped off.
Q.8 How can you explain, that following reaction is oxidation in terms of increase of oxidation number Al” Al° — A1+3 + 3e–
Answer:
Increase in oxidation number is called oxidation oxidation number of Al in creases from zero to +3 as given below Al→Al+3+3e–
Q.9 How can you prove so it is an oxidation reaction with an example that conversion of an ion to an atom is an oxidation process?
Answer:
Conversion of anion into an atom is an oxidation process.
Example:
When anions (negatively charged ions) lose electron, they are converted into atoms and oxidized.
![]()
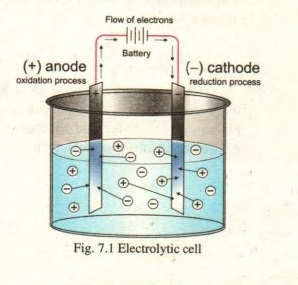
Q.10 Why does the anode carries negative charge in galvanic cell but positive charge in electrolytic cell? Justify with comments.
Answer:
In Gavanic cell, electrons are lost by the atoms at anode plate which makes it electron efficient therefore it carries negative charge. In electrolytic cell, electrons are gained by cations from anode which makes it electron deficient therefore it carries positive charge.
Q.11 Where do the electrons flow from Zn electrode in Daniel’s cell?
Answer:
In Daniel cell, the electrons takes flow from Zn electrode (anode) towards the cathode
made up of copper through the external circuit.
Q.12 Why do electrodes get their names ‘anodel and cathode in galvanic cell?
Answer:
In galvanic cell anode and cathode get their names depending upon the process taking place on them.
Anode: is an electrode where oxidation takes place
e.g. Zn → Zn+2 +2e–
Cathode: is an electrode where reduction takes place
Cu+2 +2e– → Cu
In galvanic cell, oxidation takes place at anode while reduction takes place at cathode. And oxidation always takes place at anode while reduction always takes place at cathode.
Q.13 What happens at the cathode in a galvanic cell?
Answer:
In galvanic cell, reduction takes place at the cathode as:
![]()
Q.14 Which solution is used as an electrolyte in Nelson’s cell?
Answer:
An (aqueous solution of NaCl called brine? is used as electrolyte in Nelson’s cell.
Q.15 Name the by-products produced in Nelson’s cell?
Answer:
Hydrogen gas (H2) and chlorine gas (Cl2) are the by-product of Nelson’s cell as
![]()
Q.16 Why galvanizing is done?
Answer:
The process of coating a thin layer of zinc on iron is called galvanizing. Galvanizing is done to protect the iron against corrosion even after the required coating surface is broken.
Q.17 Why an iron grill is painted frequently?
Answer:
Iron grill is painted frequently to protect it from rusting. Paint layer proctect iron from attack of moisture and oxygen.
Q.18 Why O2 is necessary for rusting?
Answer:
O2 is necessing for rusing because it acts as oxidizing agent. It accepts electrons from Fe which is covered to Fe+2 and then to Fe+3. Oxygen combines with Fe+3 to form rust (Fe2O3 H2O)
The overall cell nraction for corrosion of ions is
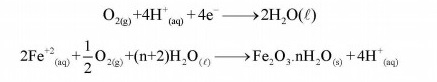
Q.19 In electroplating of chromium, which salt is used as an electrolyte?
Answer:
Chromium sulphate with few drops of H2SO4 acts as electrolyte.
Q.20 Write the redox reaction taking place during the electroplating of chromium?
Answer:
At anode:
4OH–aq → 2H2Ol +4e– +02
At cathode:
Cr–aq +3e– — >Cr(s)
Overall reaction:
Cr2(SO4)3(s)___water> Cr+3aq → 3SO-24(aq)
Q.21 In electroplating of silver, from where Ag + come and where they deposit?
Answer:
In electroplating of silver Ag+ ion come form anode while they deposit at cathode.
Q.22 What is the nature of electrode used in electroplating of chromium?
Answer:
In electroplating of chromium, anode is made of antimonial lead while the object to be electroplated acts as cathode.